Suggested wake

Want to learn Sir Thomas More about one of science's heroes from history, Henry Moseley? Moseley solved one of chemistry's greatest puzzles - determining what distinguishes elements from one another and developed a means of identifying elements supported their atomic characteristics. Sadly he lost his life fighting at Gallipoli in WWI.
Learn more almost his life and bequest by watching our online film here.
Alkaline-earth metal Metals
Alkaline Earth Metals
The alkaline-earth metal metals are the secondly most reactive family unit of elements. Be, magnesium, calcium, strontium, barium and radium are wholly clear, and achromatic. They all have low densities, thawing points and boiling points, and they tend to form solutions with a pH greater than 7.
Atomic number 4 is the only alkaline-earth metal metallic non to react with water. One of its of course occurring compounds is emerald. Calcium is needed for healthy bones, and plants need magnesium to form the chlorophyll molecules they use for photosynthesis. Strontium is important to sea life, particularly for hard corals, which use strontium to build their exoskeletons.
Radioactive Ra was discovered by Madame Curie and her hubby Pierre, and was used in incandescence-in-the-dark rouge until the 1960s!
Conspicuous Element: Calcium
 Atomic number 20 is the third most abundant metal in Earth's crust and the fifth most copious constituent. Calcium carbonate is the most inferior calcium compound happening Earth. It is found in limestone and the fossilised cadaver of prehistoric sea creatures. Other sources of atomic number 20 include gypsum, anhydrite, fluorite, and apatite.
Atomic number 20 is the third most abundant metal in Earth's crust and the fifth most copious constituent. Calcium carbonate is the most inferior calcium compound happening Earth. It is found in limestone and the fossilised cadaver of prehistoric sea creatures. Other sources of atomic number 20 include gypsum, anhydrite, fluorite, and apatite.
The name calcium derives from the Italic discussion calx, pregnant ''lime hydrate'', which was successful by heating limestone. Pure calcium was isolated in 1808 away Humphry Davy and is highly reactive. Calcium compounds have many industrialized uses, including calcium supplementation in food and medicines - atomic number 20 ions are electrolytes, with a vital theatrical role in the physiological and organic chemistry processes of organisms.
Nanoscale iron and calcium compounds that form in Alzheimer's disease
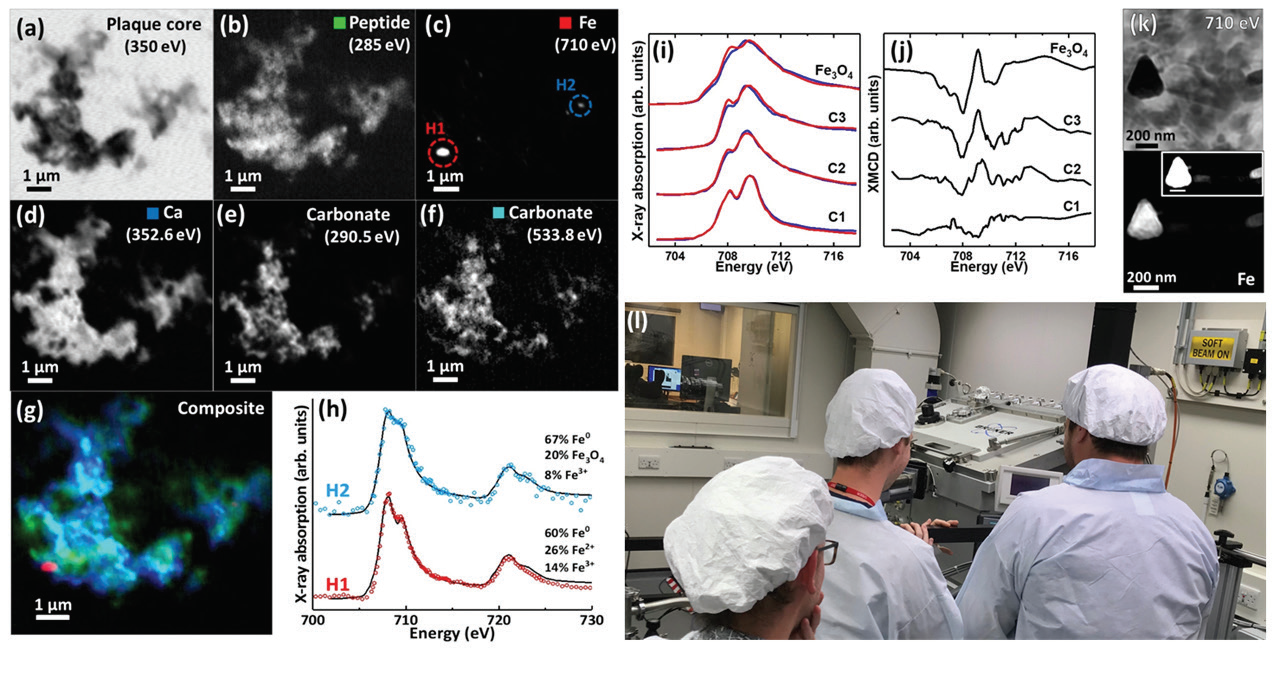 Alzheimer's is the most common form of dementia, but we don't yet understand what causes the disease, surgery how to treat it. Alzheimer's causes amyloid plaques to form in the brain that disrupt its normal function, and researchers used X-ray spectromicroscopy on the Scanning X-ray Microscopy beamline (I08) to characterise the precise distribution and chemical state of iron and atomic number 20 compounds inside amyloid plaques.
Alzheimer's is the most common form of dementia, but we don't yet understand what causes the disease, surgery how to treat it. Alzheimer's causes amyloid plaques to form in the brain that disrupt its normal function, and researchers used X-ray spectromicroscopy on the Scanning X-ray Microscopy beamline (I08) to characterise the precise distribution and chemical state of iron and atomic number 20 compounds inside amyloid plaques.
Their study revealed, in unprecedented contingent at nanoscale resolution, the properties of iron and calcium compounds in plaques from individuals who had Alzheimers. Their findings extend current hypotheses about how these and other metallic species may contribute to the development of Alzheimer's disease and could aid direct future groundbreaking diagnosis and discourse of the disease. Record more Hera.
Wherefore are natural minerals such as seashells so hard?
 Materials such as seashells, dentition and bones often have singular physical properties that are far better than those of equivalent by artificial means produced substances. Biominerals exhibit features much as complex morphologies, hierarchical structures and properties optimised for their roles that make them a unique inspiration for materials design. Researchers are particularly involved in how organisms generate materials with military strength and ruggedness from weak, brittle minerals such equally calcium carbonate and calcium phosphate.
Materials such as seashells, dentition and bones often have singular physical properties that are far better than those of equivalent by artificial means produced substances. Biominerals exhibit features much as complex morphologies, hierarchical structures and properties optimised for their roles that make them a unique inspiration for materials design. Researchers are particularly involved in how organisms generate materials with military strength and ruggedness from weak, brittle minerals such equally calcium carbonate and calcium phosphate.
An investigating using the Piping Result Pulverization Diffraction beamline (I11) provided new insights into the mechanical properties of amorphous/organic nanocomposites and determined the origin of the hardening effects of small organic molecules inside various crystals. These results are of finical implication to the mechanical properties of separate crystal biominerals and open the possibleness of using the same strategy to tailor the mechanical properties of a full range of materials. Read more here.
Task M
 Atomic number 20 carbonate has three main forms or ''polymorphs'': vaterite, calcite and aragonite. Design M was an innovative ''citizen science'' task, involving pupils from 100 schools and launched by well-known man of science Professor Alice Roberts. The initiative allowed 14-18-year-olds to contribute to cutting-edge research, take part in literal experiments and publish their results in a scientific peer-reviewed journal.
Atomic number 20 carbonate has three main forms or ''polymorphs'': vaterite, calcite and aragonite. Design M was an innovative ''citizen science'' task, involving pupils from 100 schools and launched by well-known man of science Professor Alice Roberts. The initiative allowed 14-18-year-olds to contribute to cutting-edge research, take part in literal experiments and publish their results in a scientific peer-reviewed journal.
The project aimed to gain our savvy of Ca carbonate you bet different additives touch on the different form of calcium carbonate produced. Being competent to create different types easily could be incredibly important for manufacturing.
Each participating civilis successful x samples using especially selected additives. The Diamond team analysed all of the samples in 24 hours, on the I11 beamline, to produce a giant set of results. This was the low sentence that UK schools were able to machinate and run samples at Diamond, and Project M was awarded the Royal Society of Chemistry Brainchild & Industriousness Award for 2018. Learn more here.
Discernment gypsum emergence
 Gypsum is one of three crystalline phases of calcium sulfate, alongside bassanite and anhydrite. Gypsum is one of the most common minerals on Earth, but we don't fully understand how it forms from ions in resolution. Researchers used the Small Angle Strewing and Diffraction beamline (I22) to follow the formation of gypsum in situ for the first time.
Gypsum is one of three crystalline phases of calcium sulfate, alongside bassanite and anhydrite. Gypsum is one of the most common minerals on Earth, but we don't fully understand how it forms from ions in resolution. Researchers used the Small Angle Strewing and Diffraction beamline (I22) to follow the formation of gypsum in situ for the first time.
Using Decreased and Wide Lean on X-irradiatio Scattering (SAXS and WAXS), they were able to point that gypsum forms in a four-poin process, beginning with the formation of chiseled calcium sulfate nanobricks, which self-assemble to form a crystalline structure. Time-resolved measurements allowed them to come after the ontogenesis in real time. Their results give a new insight into gypsum nucleation and maturation, which could possibly be ill-used to understand its organization in nature in the future.
Broad deposits of gypsum and anhydrite are found naturally on Earth. Bassanite is non course abundant, just has an essential role in the construction manufacture, with 100 zillion tones of IT produced annually as plaster. Evidence of natural deposits of gypsum and bassanite has also recently been found along Mars. Read more here.
Phosphate-disentangled dishwasher sparkle inevitably a chemical jazz band
 No matter how careful you are when you fill the dish washer, in that respect's no way of guaranteeing every token will fare out clean. It's specially annoying when the wine glasses come unconscious spotty, rather than sparkling. The problem is limescale - calcium carbonate - beingness left behind by the water, and it's more of a problem in hard water areas.
No matter how careful you are when you fill the dish washer, in that respect's no way of guaranteeing every token will fare out clean. It's specially annoying when the wine glasses come unconscious spotty, rather than sparkling. The problem is limescale - calcium carbonate - beingness left behind by the water, and it's more of a problem in hard water areas.
Until lately, the solvent for this return was adding a phosphate-based ''inhibitor'' to prevent the calcium build-up from forming. However, we have since discovered that although inorganic phosphate is not-toxic, it can build in the environment and induce damaging effects. High-level levels of phosphorus in rivers and lakes lede to excessive constitute maturation and algal blooms, low oxygen levels and a drop-off in biodiversity.
The EU illegal phosphates in consumer laundry detergent in 2013, and a ban along the use of phosphates in dishwashing detergent followed in 2017. Scientists are searching for less damaging chemicals that hind end do the same job, but Captain Hicks different forms of calcium carbonate, known as polymorphs, have been identified, and they don't all oppose with inhibitors in the same fashio.
A team of researchers developed a lab-based worthy dishwasher system to rapidly screen 28 chemical candidates. They used powder X-ray diffraction (PXRD) on the Small Molecule Single-Crystal Diffraction beamline (I19) to examine the atomic number 20 deposits. Their results showed that, although zero one-on-one stuff could match the performance of the phosphate-based inhibitor, two used in combining did the trick. Their combined cleaning power was habitual in a commercial message dishwasher - a truly sparkling resultant! Read many here.
And did you know?
 Happening World, our castanets are incessantly formed and reabsorbed, which gives them resistance to fractures. The calcium balance for a healthy person is (the remainder between calcium intake and excretion) is just well-nig nought. For astronauts in microgravity, still, information technology's a same different story. Their calcium balance decreases to about -250 magnesium/day, leading to swot loss and an increased hazard of kidney stones. Even though astronauts take a lot of practice session, and eat a balanced dieting, it can select some years back along Terra firma for them to recover.
Happening World, our castanets are incessantly formed and reabsorbed, which gives them resistance to fractures. The calcium balance for a healthy person is (the remainder between calcium intake and excretion) is just well-nig nought. For astronauts in microgravity, still, information technology's a same different story. Their calcium balance decreases to about -250 magnesium/day, leading to swot loss and an increased hazard of kidney stones. Even though astronauts take a lot of practice session, and eat a balanced dieting, it can select some years back along Terra firma for them to recover.
which elements are designated as the alkaline earth metals
Source: https://www.diamond.ac.uk/default/Campaigns/2019/Year-of-the-Periodic-Table/Alkaline-Earth-Metals.html